Thread replies: 5
Thread images: 1
Thread images: 1
Am I doing shit?
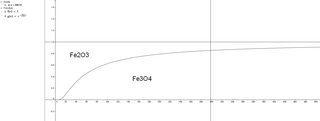
6Fe2O3 = 4Fe3O4 + O2
Q = fO2 (fugacity)
ΔrG = ΔrG° + RTlnfO2
ΔrG = 0
- ΔrG° = RTlnfO2
- ΔrG°/RT = lnfO2
exp(-ΔrG°/RT) = fO2
fO2 = exp(-391.6/8.31T)
Inducing this plot.
>>
>>8611713
Fuck off thermofag
>>
>>8611713
Your notation is messy as fuck but yeah it looks like you did everything right.
ΔG = ΔG° + RT ln(Q)
Assuming equilibrium I guess lol
0 = ΔG° + RT ln(Q)
ln(Q) = -ΔG°/RT
Q = fO2 = exp(-ΔG°/RT) = exp((-391.6) / [(8.314)(T)])
>>
>>8611713
I can't read this shit, is that equation even correct? It doesn't seem so although that may just be it being unreadable.
>>
>>8613408
It's probably correct. Look at the equilibrium established and really the only thing there is to pay attention to is the "activity" of O2, so it's safe to assume Q = fO2
Thread posts: 5
Thread images: 1
Thread images: 1